How Temperature Impacts Battery Efficiency and Performance

Battery performance is highly sensitive to temperature, especially in lithium-ion cells. At the core of battery operation are ion movements between electrodes through an electrolyte. As temperature increases, ions move more easily, reducing internal resistance and improving efficiency. When temperatures drop, the opposite occurs—ion mobility decreases, impedance rises, and efficiency falls. Temperature vs. Efficiency
Temperature vs. Efficiency
Round-trip efficiency (RTE) measures how much energy a battery can deliver compared with how much energy was required to charge it. Higher temperatures generally improve RTE because internal resistance decreases. However, gains begin to taper off beyond roughly 40 °C, and efficiency rarely exceeds about 98% for certain chemistries such as lithium iron phosphate (LFP).
More Than Just Temperature
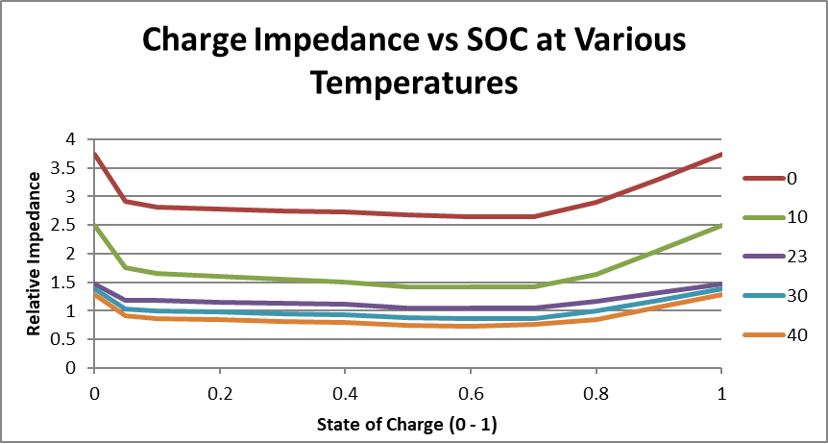
Battery impedance also varies with state of charge (SOC) and aging. Cells typically experience higher resistance at lower SOC levels, which means deeper discharge cycles can increase energy losses. Over time, resistance increases as batteries age, reducing overall efficiency.
System-Level Considerations
Battery systems typically operate within a controlled temperature range (often around 20–25 °C) using cooling equipment. While this preserves battery life and performance, cooling systems themselves consume energy, which slightly lowers total system efficiency—especially in hotter environments where more cooling is required.
The Bottom Line
Moderate operating temperatures improve battery efficiency, but maintaining those temperatures requires energy and careful system design. Battery performance ultimately depends on a combination of temperature control, operating profile, and cell chemistry.